INSIGHTS ON PHARMACEUTICAL INSPECTION
-
Advancing Drug Product Quality Through AVI And Future-Ready Infrastructure
Automated visual inspection (AVI) combines robotics, precision imaging, and sophisticated defect-recognition software to provide a scalable solution for expanding pipelines and rises in production volumes.
-
Visual Inspection Of Advanced Therapy Medicinal Products
Discover how expert database systems for inspection defect management and control help cell and gene therapies meet the requirements of USP-790 to assure they are essentially free from particulate matter.
-
What Is The Position Of Regulatory Authorities On PUPSIT?
PUPSIT and other integrity testing methods are crucial for manufacturers to reduce contamination risks and ensure drug product safety. Learn how your company can adhere to updated regulatory guidelines.
-
The Significance Of Preventive Maintenance For Product Inspection Systems
Watch to gain insight into the crucial role of maintenance in ensuring the ongoing optimal performance, reliability, and longevity of your inspection systems.
-
Integrity Testing Of Inhaler Products1/5/2026
Ensuring inhaler integrity is vital for patient safety and regulatory compliance. Discover how Vacuum decay testing provides a sensitive, non-destructive method to detect leaks and maintain sterility.
-
Guide To Product Inspection Systems In The Pharmaceutical Industry8/29/2024
Explore the critical role of inspection systems, specifically metal detectors, X-ray systems, and checkweighers, in pharmaceutical manufacturing.
-
Optimized Product Recovery Using The Drug Product Filtration System1/12/2026
Explore strategies to maximize drug product recovery during sterile filtration, minimize hold-up volume, and reduce dilution after PUPSIT to ensure higher yield, improved efficiency, and less waste.
-
Ensure Quality Control With Innovative Software7/1/2025
Ensure unmatched product quality with an X-ray system paired with an innovative software that detects contaminants and missing items in stacked packaged goods with precision.
-
OEE And Product Inspection2/13/2025
Explore the calculation of Overall Equipment Effectiveness (OEE) and the impact of product inspection equipment on OEE in the pharmaceutical industry.
PHARMACEUTICAL INSPECTION SOLUTIONS
-
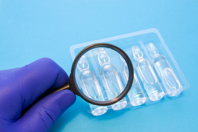
Learn how these non-destructive inspection technologies verify container closure system integrity with deterministic quantitative test methods for vials, ampoules, syringes, cartridges and auto-injectors.
-
The Thermo Scientific Versa Frame 44HB checkweigher offers dependable and accurate on-line weighing of large packages maximizing productivity and profitability.
-
V57- Customizable, precise quality control
Immediate contamination detection and precision formation inspections of rigid plastic containers.
Advanced Product Handling
Product tracking, product handling options, and intuitive software enable efficient visual inspection of every product.
Real Time Quality Control
Installed directly after product formation, the system minimizes chances that misformed or contaminated products continue to downstream processes.
Precise Package Presentation
Detect product contamination as small as 0.1 TAPPI for top-quality products.
-
The pharmaceutical industry continues to bring breakthroughs in the field of new drugs and drug delivery systems amidst the increased scrutiny of the regulators. The packaging acts as the primary sterile barrier in drug delivery systems that safeguard the drug product through its life cycle. The efficiency of the primary sterile barrier and the functional components are of utmost importance to patient safety.
-
Our Bench Vision Unit has been developed to provide maximum flexibility, with the same software and component design as our automatic visual inspection equipment used on production lines throughout the world.
- Container range: ampoules, vials, cartridges, syringes
- Inspected products: water-like, viscous, suspensions